
The electronic configuration of Sodium will be 1s2 2s2 2p6 3s1. How do you write the electron configuration for Sodium? What is the electronic configuration of Sodium 11? What is the boiling Point of Sodium in Kelvin?īoiling Point of Sodium in Kelvin is 1156 K. Melting Point of Sodium in Kelvin is 370.87 K. At room temperature Sodium is soft, silvery-white metal which can be easily cut with a knife. It is placed in group 1 of periodic table as it has a single electron in its outer most shell that it readily donates, creating a positively charged ion, the Na+ cation. What is the melting Point of Sodium in Kelvin? Sodium has an atomic number of 1 and atomic mass of 22.98. Atoms of the same element with different mass numbers are called isotopes. The number of protons and the mass number of an atom define the type of atom. What is the boiling Point of Sodium?īoiling Point of Sodium is 1156 K. Each atom has a charged sub-structure consisting of a nucleus, which is made of protons and neutrons, surrounded by electrons. A very soft, silvery white metal, the sixth most abundant element on Earth, it occurs mainly as halite, never free. 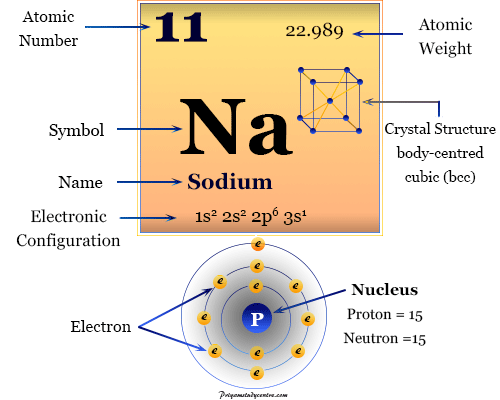
Sodium has 11 electrons out of which 1 valence electrons are present in the 3s1 outer orbitals of atom. sodium, Chemical element, one of the alkali metals, chemical symbol Na, atomic number 11. How many valence electrons does a Sodium atom have? It is located in group 1 and period 3 in the modern periodic table. Sodium is the 11 element on the periodic table. Sodium is a chemical element with the symbol Na and atomic number 11. What is the position of Sodium in the Periodic Table? Sodium is a chemical element with symbol Na and atomic number 11. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Sodium is 3s1. What is the abbreviated electronic configuration of Sodium? The electronic configuration of Sodium is 1s2 2s2 2p6 3s1. According to their increasing atomic numbers, elements are organised. What is the electronic configuration of Sodium? What are the horizontal rows on the periodic table called. Sodium Thermal Properties - Enthalpies and thermodynamics Optical Properties of Sodium Refractive IndexĪcoustic Properties of Sodium Speed of Sound Sodium Heat and Conduction Properties Thermal Conductivity Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Explore all elements Element Sodium (Na), Group 1, Atomic Number 11, s-block, Mass 22.990. Refer to table below for the Electrical properties ofSodium Electrical Conductivity Buy Sodium (Na) Element Atomic Number 11 Science Sweatshirt: Shop top fashion brands Sweatshirts at FREE DELIVERY and Returns possible on. Video References Download our free Periodic Table app for mobile phones and tablets. Hardness of Sodium - Tests to Measure of Hardness of Element Mohs Hardness Refer to below table for Sodium Physical Properties DensityĠ.968 g/cm3(when liquid at m.p density is $0.927 g/cm3)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
